Atuveciclib BAY-1143572
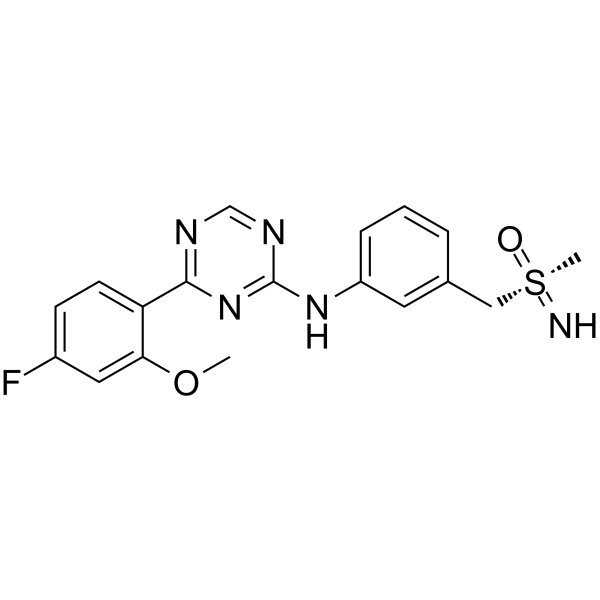
| 产品介绍 | Atuveciclib (BAY-1143572) 是一种有效的,具有口服活性的高选择性 PTEFb/CDK9 抑制剂。Atuveciclib (BAY-1143572) 抑制 CDK9/CycT1,IC50 为 13 nM。 | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 生物活性 | Atuveciclib (BAY-1143572) is a potent and highly selective, oral PTEFb/CDK9 inhibitor. Atuveciclib (BAY-1143572) inhibits CDK9/CycT1 with an IC50 of 13 nM. | ||||||||||||||||
| 体外研究 |
Positive transcription elongation factor b (PTEFb) is a heterodimer of CDK9 and one of four cyclin partners, cyclin T1, cyclin K, cyclin T2a or cyclin T2b. Atuveciclib (BAY-1143572) demonstrates potent antiproliferative activity against HeLa cells (IC50=920 nM) and MOLM-13 cells (IC50=310 nM). 西域 has not independently confirmed the accuracy of these methods. They are for reference only. | ||||||||||||||||
| 体内研究 |
In vivo efficacy studies in the MOLM-13 xenograft model in mice, Atuveciclib (BAY-1143572) demonstrates great potency and high antitumor efficacy. Daily administration of Atuveciclib (BAY-1143572) at 6.25 or 12.5 mg/kg results in a dose-dependent antitumor efficacy with a treatment-to-control (T/C) ratio of 0.64 and 0.49, respectively (p<0.001). In a separate experiment with a higher daily dose of 20 or 25 mg/kg Atuveciclib (BAY-1143572), antitumor efficacy with a T/C ratio of 0.41 and 0.31, respectively, is observed (p<0.001). The 25 mg/kg once daily dose is the maximum tolerated dose in nude mice. Furthermore, Atuveciclib (BAY-1143572) administered at 25 or 35 mg/kg, three days on / two days off, results in a T/C ratio of 0.33 and 0.20, respectively (p<0.001). Treatment with Atuveciclib (BAY-1143572) is well-tolerated, as demonstrated by less than 10 % mean body weight reduction throughout the study. In an in vivo pharmacokinetic study in rats, Atuveciclib (BAY-1143572) shows low blood clearance (CLb 1.1 L/kg per hour). 西域 has not independently confirmed the accuracy of these methods. They are for reference only. | ||||||||||||||||
| 体内研究 |
In vivo efficacy studies in the MOLM-13 xenograft model in mice, Atuveciclib (BAY-1143572) demonstrates great potency and high antitumor efficacy. Daily administration of Atuveciclib (BAY-1143572) at 6.25 or 12.5 mg/kg results in a dose-dependent antitumor efficacy with a treatment-to-control (T/C) ratio of 0.64 and 0.49, respectively (p<0.001). In a separate experiment with a higher daily dose of 20 or 25 mg/kg Atuveciclib (BAY-1143572), antitumor efficacy with a T/C ratio of 0.41 and 0.31, respectively, is observed (p<0.001). The 25 mg/kg once daily dose is the maximum tolerated dose in nude mice. Furthermore, Atuveciclib (BAY-1143572) administered at 25 or 35 mg/kg, three days on / two days off, results in a T/C ratio of 0.33 and 0.20, respectively (p<0.001). Treatment with Atuveciclib (BAY-1143572) is well-tolerated, as demonstrated by less than 10 % mean body weight reduction throughout the study. In an in vivo pharmacokinetic study in rats, Atuveciclib (BAY-1143572) shows low blood clearance (CLb 1.1 L/kg per hour). 西域 has not independently confirmed the accuracy of these methods. They are for reference only. | ||||||||||||||||
| 性状 | Solid | ||||||||||||||||
| 溶解性数据 |
In Vitro:
DMSO : ≥ 128.5 mg/mL (331.67 mM) * "≥" means soluble, but saturation unknown. 配制储备液
*
请根据产品在不同溶剂中的溶解度选择合适的溶剂配制储备液;一旦配成溶液,请分装保存,避免反复冻融造成的产品失效。 | ||||||||||||||||
| 运输条件 | Room temperature in continental US; may vary elsewhere. | ||||||||||||||||
| 储存方式 |
| ||||||||||||||||
| 参考文献 |






 浙公网安备 33010802013016号
浙公网安备 33010802013016号