Seco-DUBA,95.81%
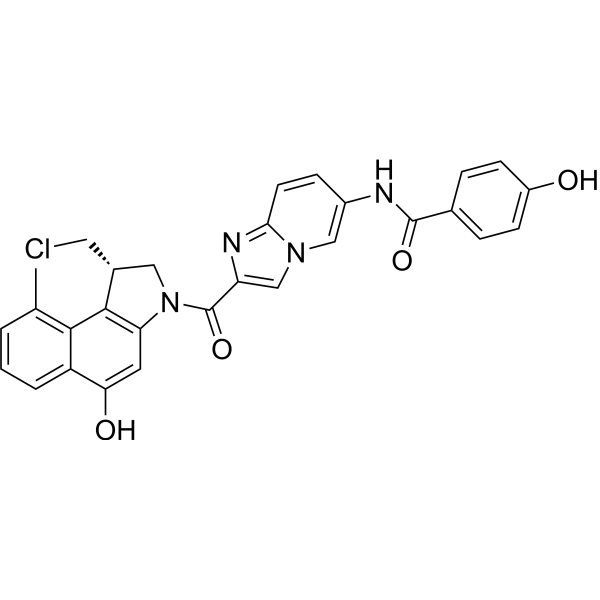
产品编号:Bellancom-132180A| CAS NO:1227961-59-2| 分子式:C29H23ClN4O4| 分子量:526.97
本网站销售的所有产品仅用于工业应用或者科学研究等非医疗目的,不可用于人类或动物的临床诊断或者治疗,非药用,非食用,
Seco-DUBA
| 产品介绍 | Seco-DUBA 是一种多卡霉素 (DUBA) 的前药,含有两个羟基,每个羟基都可以通过连接剂与抗体偶联。Seco-DUBA 可用于合成抗体-药物结合物 (ADCs)。 | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 生物活性 | Seco-DUBA is a duocarmycin (DUBA) prodrug containing two hydroxyl groups, which can each be used for coupling to an antibody via a linker. Seco-DUBA can be used in the synthesis of antibody-drug conjugates (ADCs). | ||||||||||||||||
| 体外研究 |
Seco-DUBA (SK-BR-3 cells; 0.0001 pM~0.01 nM; 144 hours) dose-dependent reduces cell viability and shows equally potent and efficacious as DUBA. 西域 has not independently confirmed the accuracy of these methods. They are for reference only. Cell Cytotoxicity Assay
|
||||||||||||||||
| 体内研究 (In Vivo) |
Seco-DUBA (89 μg/kg; i.v.) is likely converted to DUBA almost instantaneously. 西域 has not independently confirmed the accuracy of these methods. They are for reference only.
| ||||||||||||||||
| 体内研究 |
Seco-DUBA (89 μg/kg; i.v.) is likely converted to DUBA almost instantaneously. 西域 has not independently confirmed the accuracy of these methods. They are for reference only.
| ||||||||||||||||
| 体内研究 |
Seco-DUBA (89 μg/kg; i.v.) is likely converted to DUBA almost instantaneously. 西域 has not independently confirmed the accuracy of these methods. They are for reference only.
| ||||||||||||||||
| 性状 | Solid | ||||||||||||||||
| 溶解性数据 |
In Vitro:
DMSO : 100 mg/mL (189.76 mM; ultrasonic and warming and heat to 60°C) 配制储备液
*
请根据产品在不同溶剂中的溶解度选择合适的溶剂配制储备液;一旦配成溶液,请分装保存,避免反复冻融造成的产品失效。 In Vivo:
请根据您的实验动物和给药方式选择适当的溶解方案。以下溶解方案都请先按照 In Vitro 方式配制澄清的储备液,再依次添加助溶剂:
——为保证实验结果的可靠性,澄清的储备液可以根据储存条件,适当保存;体内实验的工作液,建议您现用现配,当天使用;
以下溶剂前显示的百
| ||||||||||||||||
| 运输条件 | Room temperature in continental US; may vary elsewhere. | ||||||||||||||||
| 储存方式 |
-20°C, stored under nitrogen *In solvent : -80°C, 6 months; -20°C, 1 month (stored under nitrogen) | ||||||||||||||||
| 参考文献 |






 浙公网安备 33010802013016号
浙公网安备 33010802013016号