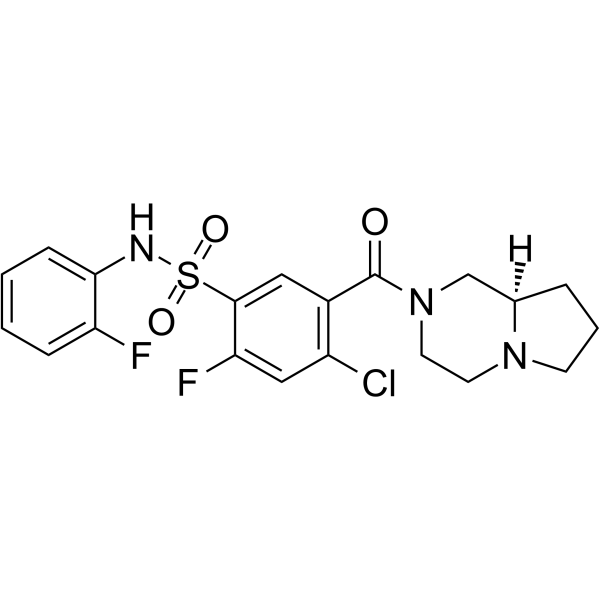
ABT-639,98.86%
产品编号:Bellancom-19721| CAS NO:1235560-28-7| 分子式:C20H20ClF2N3O3S| 分子量:455.91
本网站销售的所有产品仅用于工业应用或者科学研究等非医疗目的,不可用于人类或动物的临床诊断或者治疗,非药用,非食用,
ABT-639
| 产品介绍 | ABT-639 是一种新型选择性的 T 型 Ca2+ 通道阻滞剂。 | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 生物活性 | ABT-639 is a novel, peripherally acting, selective T-type Ca2+ channel blocker. | ||||||||||||||||
| 体外研究 | |||||||||||||||||
| 体内研究 |
ABT-639 blocks recombinant human T-type (Cav3.2) Ca2+ channels in a voltage-dependent fashion (IC50=2 μM) and attenuates low voltage-activated (LVA) currents in rat DRG neurons (IC50=8 μM). ABT-639 is significantly less active at other Ca2+ channels (e.g. Cav1.2 and Cav2.2) (IC50>30 mM). ABT-639 has high oral bioavailability (%F=73), low protein binding (88.9%) and a low brain:plasma ratio (0.05:1) in rodents. Following oral administration ABT-639 produces dose-dependent antinociception in a rat model of knee joint pain (ED50=2 mg/kg, p.o.). ABT-639 (10-100 mg/kg, p.o.) also increases tactile allodynia thresholds in multiple models of neuropathic pain (e.g. spinal nerve ligation, CCI, and vincristine-induced, and capsaicin secondary hypersensitivity). ABT-639 does not attenuate hyperalgesia in inflammatory pain models induced by complete Freund’s adjuvant or carrageenan. At higher doses (e.g. 100-300 mg/kg) ABT-639 does not significantly alter hemodynamic or psychomotor function. The antinociceptive profile of ABT-639 provides novel insights into the role of peripheral T-type (Cav3.2) channels in chronic pain states. 西域 has not independently confirmed the accuracy of these methods. They are for reference only. | ||||||||||||||||
| 体内研究 |
ABT-639 blocks recombinant human T-type (Cav3.2) Ca2+ channels in a voltage-dependent fashion (IC50=2 μM) and attenuates low voltage-activated (LVA) currents in rat DRG neurons (IC50=8 μM). ABT-639 is significantly less active at other Ca2+ channels (e.g. Cav1.2 and Cav2.2) (IC50>30 mM). ABT-639 has high oral bioavailability (%F=73), low protein binding (88.9%) and a low brain:plasma ratio (0.05:1) in rodents. Following oral administration ABT-639 produces dose-dependent antinociception in a rat model of knee joint pain (ED50=2 mg/kg, p.o.). ABT-639 (10-100 mg/kg, p.o.) also increases tactile allodynia thresholds in multiple models of neuropathic pain (e.g. spinal nerve ligation, CCI, and vincristine-induced, and capsaicin secondary hypersensitivity). ABT-639 does not attenuate hyperalgesia in inflammatory pain models induced by complete Freund’s adjuvant or carrageenan. At higher doses (e.g. 100-300 mg/kg) ABT-639 does not significantly alter hemodynamic or psychomotor function. The antinociceptive profile of ABT-639 provides novel insights into the role of peripheral T-type (Cav3.2) channels in chronic pain states. 西域 has not independently confirmed the accuracy of these methods. They are for reference only. | ||||||||||||||||
| 性状 | Solid | ||||||||||||||||
| 溶解性数据 |
In Vitro:
DMSO : 10 mg/mL (21.93 mM; Need ultrasonic and warming) 配制储备液
*
请根据产品在不同溶剂中的溶解度选择合适的溶剂配制储备液;一旦配成溶液,请分装保存,避免反复冻融造成的产品失效。 | ||||||||||||||||
| 运输条件 | Room temperature in continental US; may vary elsewhere. | ||||||||||||||||
| 储存方式 |
| ||||||||||||||||
| 参考文献 |






 浙公网安备 33010802013016号
浙公网安备 33010802013016号