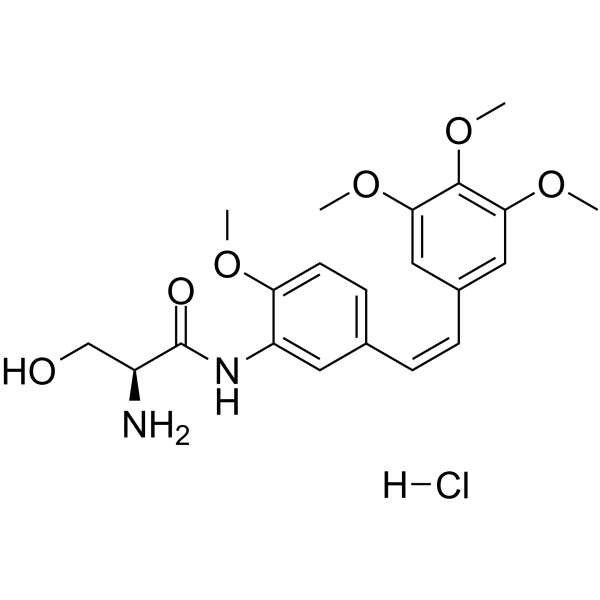
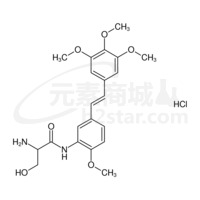
Ombrabulin hydrochloride AVE8062 hydrochloride; AC7700 hydrochloride,99.43%
产品编号:Bellancom-18256| CAS NO:253426-24-3| 分子式:C21H27ClN2O6| 分子量:438.90
本网站销售的所有产品仅用于工业应用或者科学研究等非医疗目的,不可用于人类或动物的临床诊断或者治疗,非药用,非食用,
Ombrabulin hydrochloride AVE8062 hydrochloride; AC7700 hydrochloride
| 产品介绍 | Ombrabulin hydrochloride 是 CA-4 磷酸酯的衍生物,选择性破坏内皮细胞的微管蛋白细胞骨架,具有抗血管作用。 | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 生物活性 | Ombrabulin hydrochloride is a derivative of CA-4 phosphate, which is known to exhibit antivascular effects through selective disruption of the tubulin cytoskeleton of endothelial cells. | ||||||||||||||||
| 体外研究 |
The effect of Ombrabulin (AVE8062) on endothelial or tumor cell viability is examined using the MTT assay. The IC50 of Ombrabulin for the mouse mesenteric endothelial cells (MMEC) is 10 nM and ranges between 7 and 20 nM for the tumor cell lines (HeyA8, SKOV3ip1, and HeyA8-MDR). Comparative analysis of the nonlinear least-squares regression of the dose-response curves for each agent alone and combination Ombrabulin /Docetaxel show a significantly lower IC50 than either agent alone (P<0.005, all cell lines). The cytotoxicity of Docetaxel is 2- to 4-fold greater in combination with Ombrabulin for the endothelial and tumor cells compared with Docetaxel alone. 西域 has not independently confirmed the accuracy of these methods. They are for reference only. | ||||||||||||||||
| 体内研究 |
Before performing therapy experiments, the tolerability of various doses of Ombrabulin (AVE8062) ranging from 10 to 100 mg/kg is tested given twice weekly via i.v., i.p., or s.c. routes in nude mice (n=3 per group). The i.v. and s.c. routes are not pursued further due to problems with skin or tail vein necrosis. The i.p. route is well tolerated with doses up to 100 mg/kg. Next, preliminary experiments are done to determine the lowest dose for in vivo therapeutic efficacy. Starting 7 days after tumor cell injection, nude mice (n=5 per group) bearing HeyA8 ovarian cancer cells are treated with either vehicle or Ombrabulin 10, 30, 50, and 100 mg/kg twice weekly i.p. for 3 weeks. There is 65% reduction in tumor weight in the 30 mg/kg group compared with the vehicle control group (P<0.02). The 10 mg/kg dose is not effective. The antitumor effects at doses >30 mg/kg are not significantly better; therefore, the 30 mg/kg dose is selected for subsequent therapy experiments. 西域 has not independently confirmed the accuracy of these methods. They are for reference only. | ||||||||||||||||
| 体内研究 |
Before performing therapy experiments, the tolerability of various doses of Ombrabulin (AVE8062) ranging from 10 to 100 mg/kg is tested given twice weekly via i.v., i.p., or s.c. routes in nude mice (n=3 per group). The i.v. and s.c. routes are not pursued further due to problems with skin or tail vein necrosis. The i.p. route is well tolerated with doses up to 100 mg/kg. Next, preliminary experiments are done to determine the lowest dose for in vivo therapeutic efficacy. Starting 7 days after tumor cell injection, nude mice (n=5 per group) bearing HeyA8 ovarian cancer cells are treated with either vehicle or Ombrabulin 10, 30, 50, and 100 mg/kg twice weekly i.p. for 3 weeks. There is 65% reduction in tumor weight in the 30 mg/kg group compared with the vehicle control group (P<0.02). The 10 mg/kg dose is not effective. The antitumor effects at doses >30 mg/kg are not significantly better; therefore, the 30 mg/kg dose is selected for subsequent therapy experiments. 西域 has not independently confirmed the accuracy of these methods. They are for reference only. | ||||||||||||||||
| 性状 | Solid | ||||||||||||||||
| 溶解性数据 |
In Vitro:
H2O : 20 mg/mL (45.57 mM; Need ultrasonic) 配制储备液
*
请根据产品在不同溶剂中的溶解度选择合适的溶剂配制储备液;一旦配成溶液,请分装保存,避免反复冻融造成的产品失效。 | ||||||||||||||||
| 运输条件 | Room temperature in continental US; may vary elsewhere. | ||||||||||||||||
| 储存方式 |
4°C, sealed storage, away from moisture *In solvent : -80°C, 6 months; -20°C, 1 month (sealed storage, away from moisture) | ||||||||||||||||
| 参考文献 |






 浙公网安备 33010802013016号
浙公网安备 33010802013016号