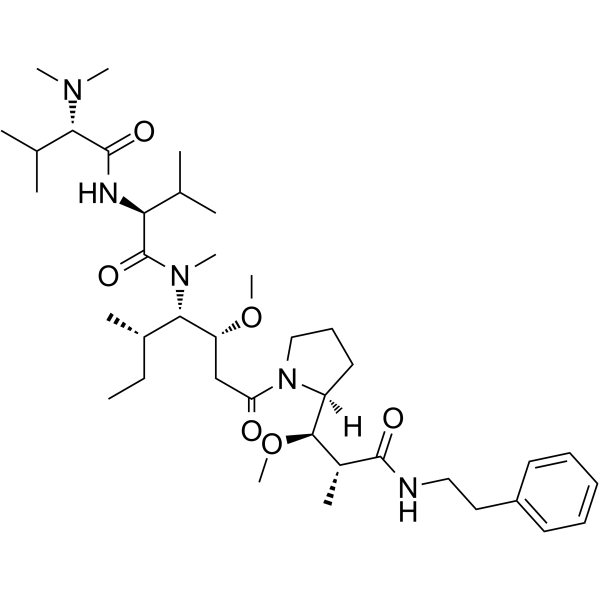
Soblidotin Auristatin PE; TZT-1027,99.64%
产品编号:Bellancom-14672| CAS NO:149606-27-9| 分子式:C39H67N5O6| 分子量:701.98
本网站销售的所有产品仅用于工业应用或者科学研究等非医疗目的,不可用于人类或动物的临床诊断或者治疗,非药用,非食用,
Soblidotin Auristatin PE; TZT-1027
| 产品介绍 | Soblidotin (Auristatin PE) 是一种新型合成的 Dolastatin 10衍生物,抑制剂微管 (tubulin) 聚合。 | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 生物活性 | Soblidotin (Auristatin PE) is a novel synthetic Dolastatin 10 derivative and inhibitor of tubulin polymerization. | ||||||||||||||||
| 体外研究 |
Soblidotin (Auristatin PE) is a novel synthetic dolastatin 10 derivative that inhibits tubulin polymerization. Soblidotin (Auristatin PE) exhibits antitumor activity against p-glycoprotein-overexpressing cell lines established from colon cancer H116 and breast cancer-resistant protein-positive cell lines established from lung cancer PC-6, and is more potent than Vincristine, Paclitaxel, and Docetaxel against these cell lines. Soblidotin (Auristatin PE) is a synthetic analog of dolastatin 10 which inhibits the growth of several tumoral cell lines and induces caspase-3-dependent apoptosis. Soblidotin (Auristatin PE) also shows antitumoral activity in Vincristine-, Docetaxel-, and Paclitaxel-resistant tumors, which makes it a potential chemotherapy drug for use in tumors which do not respond to other microtubule inhibitors. 西域 has not independently confirmed the accuracy of these methods. They are for reference only. | ||||||||||||||||
| 体内研究 |
Intravenous injection of Auristatin PE (TZT-1027) has been shown to potently inhibit the growth of P388 leukemic cells and several solid tumors in mice, and to prolong the survival of the animals, and its antitumor efficacy has been shown to be superior or comparable to that of the reference agents Dolastatin 10, Cisplatin, Vincristine, and 5-Fluorouracil. Furthermore, in xenograft models, Auristatin PE reduces intratumoral blood perfusion 1 to >24 h after its administration, thereby producing hemorrhagic necrosis of the tumors. Auristatin PE (Soblidotin) shows antivascular effects in tumoral models overexpressing VEGF and in murine colon tumors, with an increase in vascular permeability, vessel closure, and widespread hemorrhage. Mice bearing subcutaneous HT-29 tumors (200 mm3) are dosed every 7 days with Auristatin PE (0.5 or 1.0 mg/kg) for a total of four cycles. Under such conditions, Auristatin PE (TZT-1027) inhibits the growth of HT-29 xenografts in a dose-dependent manner. Coadministration of Auristatin PE does not interfere with the PD184352-induced suppression of ERK1/2 phosphorylation. Immunostaining for Ki-67 as a marker for proliferating cells confirmed that the number of such cells in tumor sections is decreased greatly at 24 hours after the initial dosing with PD184352 compared with that apparent for vehicle-treated tumors. Auristatin PE treatment alone increases the number of TUNEL-positive cells in HT-29 xenografts by 24 hours in a dose-dependent manner, and this effect is enhanced by coadministration of PD184352. 西域 has not independently confirmed the accuracy of these methods. They are for reference only. | ||||||||||||||||
| 体内研究 |
Intravenous injection of Auristatin PE (TZT-1027) has been shown to potently inhibit the growth of P388 leukemic cells and several solid tumors in mice, and to prolong the survival of the animals, and its antitumor efficacy has been shown to be superior or comparable to that of the reference agents Dolastatin 10, Cisplatin, Vincristine, and 5-Fluorouracil. Furthermore, in xenograft models, Auristatin PE reduces intratumoral blood perfusion 1 to >24 h after its administration, thereby producing hemorrhagic necrosis of the tumors. Auristatin PE (Soblidotin) shows antivascular effects in tumoral models overexpressing VEGF and in murine colon tumors, with an increase in vascular permeability, vessel closure, and widespread hemorrhage. Mice bearing subcutaneous HT-29 tumors (200 mm3) are dosed every 7 days with Auristatin PE (0.5 or 1.0 mg/kg) for a total of four cycles. Under such conditions, Auristatin PE (TZT-1027) inhibits the growth of HT-29 xenografts in a dose-dependent manner. Coadministration of Auristatin PE does not interfere with the PD184352-induced suppression of ERK1/2 phosphorylation. Immunostaining for Ki-67 as a marker for proliferating cells confirmed that the number of such cells in tumor sections is decreased greatly at 24 hours after the initial dosing with PD184352 compared with that apparent for vehicle-treated tumors. Auristatin PE treatment alone increases the number of TUNEL-positive cells in HT-29 xenografts by 24 hours in a dose-dependent manner, and this effect is enhanced by coadministration of PD184352. 西域 has not independently confirmed the accuracy of these methods. They are for reference only. | ||||||||||||||||
| 性状 | Solid | ||||||||||||||||
| 溶解性数据 |
In Vitro:
DMSO : 100 mg/mL (142.45 mM; Need ultrasonic) H2O : < 0.1 mg/mL (insoluble) 配制储备液
*
请根据产品在不同溶剂中的溶解度,选择合适的溶剂配制储备液;该产品在溶液状态不稳定,建议您现用现配,即刻使用。 In Vivo:
请根据您的实验动物和给药方式选择适当的溶解方案。以下溶解方案都请先按照 In Vitro 方式配制澄清的储备液,再依次添加助溶剂:
——为保证实验结果的可靠性,澄清的储备液可以根据储存条件,适当保存;体内实验的工作液,建议您现用现配,当天使用;
以下溶剂前显示的百
*以上所有助溶剂都可在 西域 网站选购。
| ||||||||||||||||
| 运输条件 | Room temperature in continental US; may vary elsewhere. | ||||||||||||||||
| 储存方式 |
*该产品在溶液状态不稳定,建议您现用现配,即刻使用。 | ||||||||||||||||
| 参考文献 |
|






 浙公网安备 33010802013016号
浙公网安备 33010802013016号