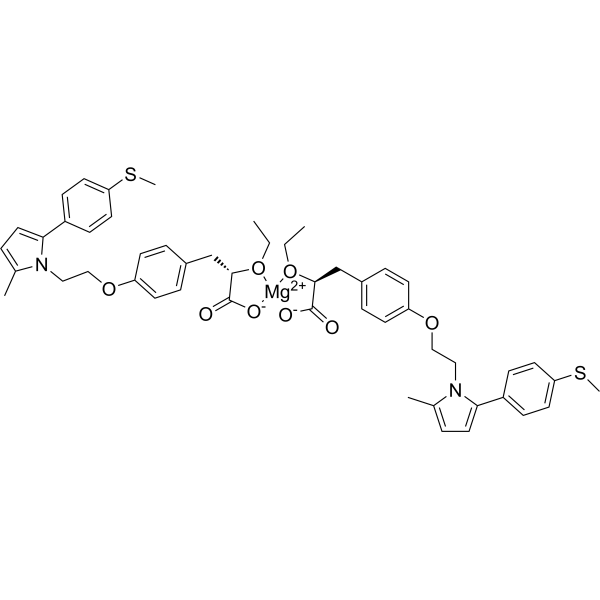
Saroglitazar magnesium 沙罗格列扎镁盐,98.00%
产品编号:Bellancom-19937A| CAS NO:1639792-20-3| 分子式:C50H56MgN2O8S2| 分子量:901.42
本网站销售的所有产品仅用于工业应用或者科学研究等非医疗目的,不可用于人类或动物的临床诊断或者治疗,非药用,非食用,
Saroglitazar magnesium 沙罗格列扎镁盐
| 产品介绍 | Saroglitazar magnesium 是一种新型的过氧化物酶体增殖物活化受体PPAR的激动剂,具有显著PPARα活性和中度PPARγ活性,在HepG2细胞中的EC50值分别为0.65 pM and 3 nM。 | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 生物活性 | Saroglitazar magnesium is a novel peroxisome proliferator-activated receptor (PPAR) agonist with predominant PPARα and moderate PPARγ activity with EC50 values of 0.65 pM and 3 nM in HepG2 cells, respectively. | ||||||||||||||||
| 体外研究 | |||||||||||||||||
| 体内研究 |
In db/db mice, 12-day treatment with Saroglitazar (0.01-3 mg/kg per day, orally) causes dose-dependent reductions in serum triglycerides (TG), free fatty acids (FFA), and glucose. The ED50 for these effects is found to be 0.05, 0.19, and 0.19 mg/kg, respectively with AUC-glucose following oral glucose administration (59%) at 1 mg/kg dose. A 90-day repeated dose comparative study in Wistar rats and marmosets confirms efficacy (TG lowering) potential of Saroglitazar and has indicated low risk of PPAR-associated side effects in humans. Based on efficacy and safety profile, Saroglitazar appears to have good potential as novel therapeutic agent for treatment of dyslipidemia and diabetes. 西域 has not independently confirmed the accuracy of these methods. They are for reference only. | ||||||||||||||||
| 体内研究 |
In db/db mice, 12-day treatment with Saroglitazar (0.01-3 mg/kg per day, orally) causes dose-dependent reductions in serum triglycerides (TG), free fatty acids (FFA), and glucose. The ED50 for these effects is found to be 0.05, 0.19, and 0.19 mg/kg, respectively with AUC-glucose following oral glucose administration (59%) at 1 mg/kg dose. A 90-day repeated dose comparative study in Wistar rats and marmosets confirms efficacy (TG lowering) potential of Saroglitazar and has indicated low risk of PPAR-associated side effects in humans. Based on efficacy and safety profile, Saroglitazar appears to have good potential as novel therapeutic agent for treatment of dyslipidemia and diabetes. 西域 has not independently confirmed the accuracy of these methods. They are for reference only. | ||||||||||||||||
| 性状 | Solid | ||||||||||||||||
| 溶解性数据 |
In Vitro:
DMSO : 125 mg/mL (138.67 mM; Need ultrasonic) 配制储备液
*
请根据产品在不同溶剂中的溶解度选择合适的溶剂配制储备液;一旦配成溶液,请分装保存,避免反复冻融造成的产品失效。 In Vivo:
请根据您的实验动物和给药方式选择适当的溶解方案。以下溶解方案都请先按照 In Vitro 方式配制澄清的储备液,再依次添加助溶剂:
——为保证实验结果的可靠性,澄清的储备液可以根据储存条件,适当保存;体内实验的工作液,建议您现用现配,当天使用;
以下溶剂前显示的百
| ||||||||||||||||
| 运输条件 | Room temperature in continental US; may vary elsewhere. | ||||||||||||||||
| 储存方式 |
4°C, sealed storage, away from moisture *In solvent : -80°C, 6 months; -20°C, 1 month (sealed storage, away from moisture) | ||||||||||||||||
| 参考文献 |






 浙公网安备 33010802013016号
浙公网安备 33010802013016号