Rabacfosadine GS-9219; VDC-1101,99.26%
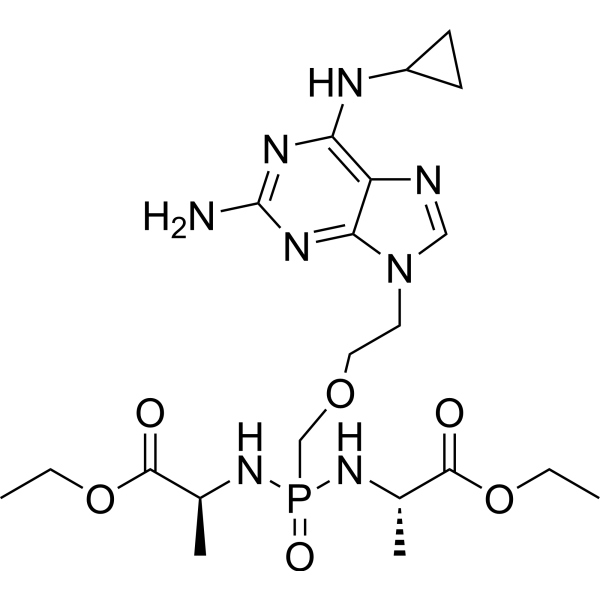
产品编号:Bellancom-13640| CAS NO:859209-74-8| 分子式:C21H35N8O6P| 分子量:526.53
本网站销售的所有产品仅用于工业应用或者科学研究等非医疗目的,不可用于人类或动物的临床诊断或者治疗,非药用,非食用,
Rabacfosadine GS-9219; VDC-1101
| 产品介绍 | Rabacfosadine (GS-9219) 是核苷类似物 PMEG 的新型前体,用作优先靶向淋巴细胞的细胞毒性剂。 | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 生物活性 | Rabacfosadine (GS-9219), a novel proagent of the nucleotide analogue PMEG, is designed as a cytotoxic agent that preferentially targets lymphoid cells. | ||||||||||||||||
| 体外研究 |
In lymphocytes, Rabacfosadine (GS-9219) is converted to its active metabolite, 9-(2-phosphonylmethoxyethyl)guanine (PMEG) diphosphate, via enzymatic hydrolysis, deamination, and phosphorylation. GS-9219 has substantial antiproliferative activity against activated lymphocytes and hematopoietic tumor cell lines. The ability of Rabacfosadine to inhibit the proliferation of activated lymphocytes and of tumor cells of hematopoietic origin is investigated. Rabacfosadine inhibits the proliferation of mitogen-stimulated T and B lymphocytes with EC50 values of 135 and 42 nM, respectively, as determined by BrdUrd incorporation. To compare the activity of GS-9219 in dividing and nondividing cells, Rabacfosadine is evaluated in these populations using a metabolism-based sodium XTT assay instead of BrdUrd assays. Results from the XTT assay shows a 127-fold difference between the EC50 values of Rabacfosadine in quiescent (EC50=17.2 μM) and proliferating (EC50=135 nM) cells. These results indicate a substantial selectivity of Rabacfosadine toward actively replicating lymphoblasts. 西域 has not independently confirmed the accuracy of these methods. They are for reference only. | ||||||||||||||||
| 体内研究 |
Rabacfosadine (RAB) has substantial single-agent activity in dogs with lymphoma, and a different mechanism of action than Doxorubicin (DOX). Open-label, multicenter prospective clinical trial. Dogs receive alternating Rabacfosadine (1.0 mg/kg IV weeks 0, 6, 12) and Doxorubicin (30 mg/m2 IV weeks 3, 9, 15). Dogs that achieved complete response (CR) are followed by monthly evaluations. Complete clinicopathological evaluation and assessment of remission and adverse event (AEs) are performed every 21 days. Acute AEs, occurring within 21 days after administration of the first dose of each agent, are compared between Rabacfosadine and Doxorubicin in 46 dogs receiving at least 1 dose of each agent. 西域 has not independently confirmed the accuracy of these methods. They are for reference only. | ||||||||||||||||
| 体内研究 |
Rabacfosadine (RAB) has substantial single-agent activity in dogs with lymphoma, and a different mechanism of action than Doxorubicin (DOX). Open-label, multicenter prospective clinical trial. Dogs receive alternating Rabacfosadine (1.0 mg/kg IV weeks 0, 6, 12) and Doxorubicin (30 mg/m2 IV weeks 3, 9, 15). Dogs that achieved complete response (CR) are followed by monthly evaluations. Complete clinicopathological evaluation and assessment of remission and adverse event (AEs) are performed every 21 days. Acute AEs, occurring within 21 days after administration of the first dose of each agent, are compared between Rabacfosadine and Doxorubicin in 46 dogs receiving at least 1 dose of each agent. 西域 has not independently confirmed the accuracy of these methods. They are for reference only. | ||||||||||||||||
| 性状 | Solid | ||||||||||||||||
| 溶解性数据 |
In Vitro:
DMSO : 100 mg/mL (189.92 mM; Need ultrasonic) 配制储备液
*
请根据产品在不同溶剂中的溶解度选择合适的溶剂配制储备液;一旦配成溶液,请分装保存,避免反复冻融造成的产品失效。 In Vivo:
请根据您的实验动物和给药方式选择适当的溶解方案。以下溶解方案都请先按照 In Vitro 方式配制澄清的储备液,再依次添加助溶剂:
——为保证实验结果的可靠性,澄清的储备液可以根据储存条件,适当保存;体内实验的工作液,建议您现用现配,当天使用;
以下溶剂前显示的百
| ||||||||||||||||
| 运输条件 | Room temperature in continental US; may vary elsewhere. | ||||||||||||||||
| 储存方式 |
| ||||||||||||||||
| 参考文献 |
|






 浙公网安备 33010802013016号
浙公网安备 33010802013016号