GSK3186899 DDD-853651,98.61%
产品编号:Bellancom-112622| CAS NO:1972617-87-0| 分子式:C19H28F3N7O3S| 分子量:491.53
本网站销售的所有产品仅用于工业应用或者科学研究等非医疗目的,不可用于人类或动物的临床诊断或者治疗,非药用,非食用,
GSK3186899 DDD-853651
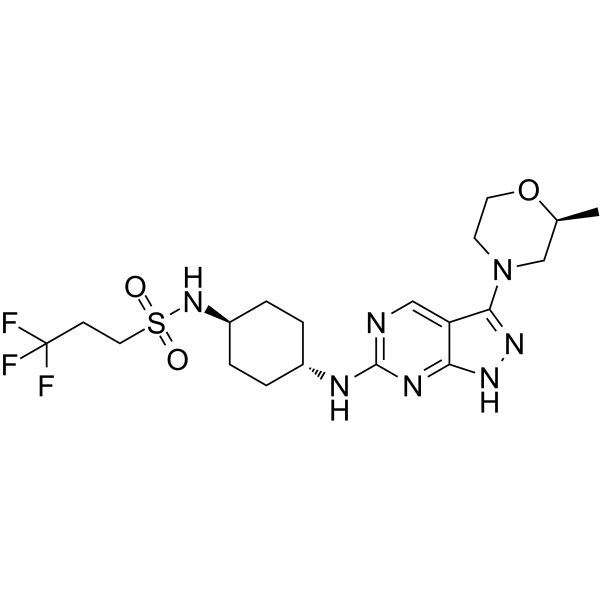
| 产品介绍 | GSK3186899 (DDD-853651) 是 CRK12 的一个抑制剂,其对 L. donovani 的 EC50 值为 1.4 μM。 | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 生物活性 | GSK3186899 (DDD-853651) is an inhibitor of cdc-2-related kinase 12 (CRK12), with an EC50 of 1.4 μM for L. donovani in an intra-macrophage assay. | ||||||||||||||||
| 体外研究 |
GSK3186899 (Compound 7) is active against L. donovani in an intra-macrophage assay with an EC50 value of 1.4 μM, and shows good selectivity against mammalian THP-1 host cells (EC50 value>50 μM). This is not as potent as reported data for amphotericin B (EC50 value of 0.07 μM in the intra-macrophage assay), but is comparable to the clinically used drugs miltefosine and paromomycin (EC50 values of 0.9 μM and 6.6 μM, respectively). GSK3186899 is also active in cidal axenic amastigote assay (EC50 value of 0.1 μM). At a concentration of 0.2 μM, GSK3186899 is cytocidal at 96 h; increasing the concentration to 1.8 μM reduced this time to 48 h. GSK3186899 demonstrates a less than 10-fold variation in potency against a panel of Leishmania-derived lines. GSK3186899 is also more active in a panel of Leishmania lines using human peripheral blood mononuclear cells as the host cells. 西域 has not independently confirmed the accuracy of these methods. They are for reference only. | ||||||||||||||||
| 体内研究 |
In the mouse model of infection, GSK3186899 demonstrates comparable activity to the front-line drug miltefosine, reducing parasite levels by 99% when dosed orally twice a day for 10 days at 25 mg/kg. The efficacy of treatment is dependent on dose, frequency, and duration (10 days better than 5). The non-clinical safety data for GSK3186899 suggests a suitable therapeutic window for progression into regulatory preclinical studies. Non-GLP preclinical assessment of cardiovascular effects and genotoxicity does not reveal any issues that would prevent further development. In addition, there are no notable adverse effects in a rat seven-day repeat-dose oral toxicity study with respect to clinical chemistry and histopathology at all doses tested. Both the in vivo efficacy and safety profile of GSK3186899 support progression to definitive safety studies. 西域 has not independently confirmed the accuracy of these methods. They are for reference only. | ||||||||||||||||
| 体内研究 |
In the mouse model of infection, GSK3186899 demonstrates comparable activity to the front-line drug miltefosine, reducing parasite levels by 99% when dosed orally twice a day for 10 days at 25 mg/kg. The efficacy of treatment is dependent on dose, frequency, and duration (10 days better than 5). The non-clinical safety data for GSK3186899 suggests a suitable therapeutic window for progression into regulatory preclinical studies. Non-GLP preclinical assessment of cardiovascular effects and genotoxicity does not reveal any issues that would prevent further development. In addition, there are no notable adverse effects in a rat seven-day repeat-dose oral toxicity study with respect to clinical chemistry and histopathology at all doses tested. Both the in vivo efficacy and safety profile of GSK3186899 support progression to definitive safety studies. 西域 has not independently confirmed the accuracy of these methods. They are for reference only. | ||||||||||||||||
| 性状 | Solid | ||||||||||||||||
| 溶解性数据 |
In Vitro:
DMSO : 250 mg/mL (508.62 mM; Need ultrasonic) 配制储备液
*
请根据产品在不同溶剂中的溶解度选择合适的溶剂配制储备液;一旦配成溶液,请分装保存,避免反复冻融造成的产品失效。 In Vivo:
请根据您的实验动物和给药方式选择适当的溶解方案。以下溶解方案都请先按照 In Vitro 方式配制澄清的储备液,再依次添加助溶剂:
——为保证实验结果的可靠性,澄清的储备液可以根据储存条件,适当保存;体内实验的工作液,建议您现用现配,当天使用;
以下溶剂前显示的百
| ||||||||||||||||
| 运输条件 | Room temperature in continental US; may vary elsewhere. | ||||||||||||||||
| 储存方式 |
| ||||||||||||||||
| 参考文献 |






 浙公网安备 33010802013016号
浙公网安备 33010802013016号